
ISCD Learning
Catalog Advanced Search
-
Includes Multiple Live Events. The next is on 12/07/2024 at 10:00 AM (EST)
Course Date: December 7 & 8, 2024. The core elements of this program focus on performing quality DXA scans, dissecting the data and applying this knowledge to a technologist’s role in patient care. This course, which focuses on quality DXA, is innovative, interactive and informative. The course emphasizes technical excellence in the performance, analysis and troubleshooting of bone densitometry scans. It provides standard practices and applications to raise the quality of DXA scan procedures and their impact on patient care. The program is designed to enhance awareness of the technical expertise involved in performing bone densitometry and applying protocols to address the everyday challenges. Attendees with come away with a strong sense of the considerable impact that a technologist plays in the delivery of quality DXA, and how that influences patient management.
Course Date
December 7 & 8, 2024.
Course Description
The core elements of this program focus on performing quality DXA scans, dissecting the data and applying this knowledge to a technologist’s role in patient care. This course, which focuses on quality DXA, is innovative, interactive and informative. The course emphasizes technical excellence in the performance, analysis and troubleshooting of bone densitometry scans. It provides standard practices and applications to raise the quality of DXA scan procedures and their impact on patient care. The program is designed to enhance awareness of the technical expertise involved in performing bone densitometry and applying protocols to address the everyday challenges. Attendees with come away with a strong sense of the considerable impact that a technologist plays in the delivery of quality DXA, and how that influences patient management.
Target Audience
The course is most appropriate for those involved in the performance of DXA scans and open to any health professional performing these scans.
Learning Objectives
After attending the course, participants should be better prepared to:
- Design and implement quality control and assurance practices.
- Perform a precision assessment and appreciate the requirement for clinical care.
- Identify the anatomy relevant to clinical DXA scanning.
- Implement consistent and appropriate technical acquisition and analysis techniques.
- Perform a fracture risk assessment using FRAX.
- Provide DXA interpreters sufficient information for a high-quality DXA report.
- Discuss supplemental software and their application.
- Demonstrate how to access features within the DXA software.
- Understand how specific medical conditions and medications affect bone density.
- Critically evaluate DXA scans to identify acquisition and analysis errors and implement resolution methods.
- Recognize how technical errors impact clinical interpretation and patient care.
Method of Participation
This activity will be offered through zoom, allowing the learner to watch and ask questions in a live format on December 7 & 8, 2024. Learners will be asked to complete a post-course survey to indicate what they will change in their practice as a result of this activity.
Registration Fee
Cancellation Policy
Attendee Cancellation Policy All changes in registration must be made in writing to education@iscd.org. Cancellations received 30 days prior to the start of the program are eligible for a full refund minus a $50 administrative fee. Cancellations received less than 30 days prior to the start of the course will not be refunded.
Course Changes or Cancellations ISCD reserves the right to cancel the program, change dates and/or meeting location. If a program is canceled, all program fees will be refunded automatically unless the registered participant elects to transfer to another program. Participants will be notified of any changes within 21 days of the course start date.
ASRT Credit Designation
This activity has been approved by ASRT for 13.0 Category A continuing education credits.
ASRT Credit Notice
This course is a renewal. If you have taken the course since June 2022, you will not be able to claim ASRT again for this learning activity. If you have taken prior to June 2022, you can take this course again for credit.
Deadline to Claim Credit
Participants will have 90 days from the activity end date to complete the activity survey and claim continuing education credit.
Schedule - Day 1 - All times Eastern (NYC/USA)
Schedule - Day 2 - All times Eastern (NYC/USA)

Anita Colquhoun, RTNM, MRT(N), CDT
Charge Technologist
Women's College Hospital
Anita Colquhoun M.R.T.(N), CDT is the charge technologist in the Centre for Osteoporosis & Bone Health at the Women’s College Hospital in Toronto. Anita is a Nuclear Medical Technologist specialized in Bone Mineral Density (BMD). She has been working in the field of BMD since the early 1990s. Anita is an active member ISCD Technologist Faculty since 2000. She has served 2 terms on the Governing Board of the ISCD and is currently serving her third term as the Executive Secretary to the Board. As well as Chairing then Annual Meeting Committee from 2012-2016 Anita served and is still serving on several other committees within the ISCD. She has participated in a variety of research studies and has co-authored abstracts and papers. Anita has also co-written an on-line beginner BMD course as well as developed a competency profile and best practise in BMD for the Ontario Association of Medical Radiation Sciences.

Diane Krueger, BS, CBDT
Researcher
Osteoporosis Clinical Research Program - University of Wisconsin, Madison
Diane Krueger received her Bachelor of Science degree at the University of Wisconsin-Madison. She is an ISCD-certified clinical densitometrist and a certified clinical research coordinator through the Association of Clinical Research Professionals. She has been program manager of the University of Wisconsin Osteoporosis Clinical Research Program since its inception in 1993. Ms. Krueger has extensive clinical research experience in osteoporosis and bone densitometry, having coordinated multiple industry and investigator-initiated studies. In collaboration with the UW Osteoporosis Program, she has published over 90 manuscripts and authored or presented over 200 abstracts. Her service with ISCD has included serving as Technologist Bone Densitometry Course faculty since 2006 and chairing the related Update Annual Meeting Committees. In her leadership capacity, she is currently Education Council chair and serves on the Executive Committee and Board. Additionally, she previously held several officer positions including Secretary and four Presidential seats.

Greg Yardley
Sr. Medical Radiology Technologist and Researcher
NASA Bone Mineral Laboratory
Greg Yardley works as a SR. Medical Radiology Technologist and researcher in the NASA Bone Mineral Laboratory and NASA/JSC medical clinic for KBR in the government solutions division. His job requirements are performing and analyzing, Dual Energy x-ray absorptiometry (DXA) exams on both retired and active Astronauts. He also extracts, records, aids in bone density interpretation and distributes bone data from Dual Energy xray absorptiometry (DXA), Computed Tomography (CT), Magnetic resonance imaging devises. Greg also performs diagnostic medical x-rays for the NASA/JSC Flight medicine and occupational health clinic.
Greg began his healthcare career as an Emergency Medical Technician (EMT). He then furthered his medical education and received his certification as a registered radiology technologist. Upon completion of his Radiological Science courses. Greg has worked as a Radiology technologist in a Trauma level 1 Hospital, Level III hospital, urgent care, and multi-specialty clinic. At the time of his graduation Greg had also received extensive training not only in X-ray but also DXA, CT, and Ultrasound. In 2011 just previous to his current job Greg got a job as a Bone Density Technologist at a multi-specialty medical clinic where he performed and analyzed over 3000 DXA scans a year on every type of patient imaginable while also running the day-to-day operations of the Osteoporosis center.

Statement of Independence
The ISCD maintains a policy on the use of commercial support, which ensures that all educational activities sponsored by the ISCD provide in-depth presentations that are fair, balanced, independent, and scientifically rigorous. ISCD requires faculty, planners, managers, and other individuals and their spouse/life partner who are in a position to control the content of this activity to disclose any real or apparent conflict of interest they may have as related to the content of this activity. All identified conflicts of interest are thoroughly vetted by ISCD for fair balance, scientific objectivity of studies mentioned in the materials or used as the basis for content, and appropriateness of patient care recommendations. Individual disclosures are included in the course material. No Commercial Support has been provided for this activity
Disclaimer
The information and suggestions presented at the courses, seminars and other programs sponsored by ISCD and other collaborating societies are subject to change and therefore should serve only as a foundation for further investigation and study. Any forms presented at our seminars or programs are samples only and are not necessarily authoritative. All information, procedures, and forms contained or used in such seminars or programs should serve only as a guide for use in specific situations.
Disclosure of Unlabeled Use
This educational activity may contain a discussion of published and/or investigational uses of agents that are not indicated by the FDA. ISCD does not recommend the use of any agent outside of the labeled indications. The opinions expressed in this educational activity are those of the faculty and do not necessarily represent the views of any organization associated with this activity. Please refer to the official prescribing information for each product for a discussion of approved indications, contraindications, and warnings.
Privacy & Confidentiality Statement
The ISCD will record the learner’s personal information as provided on evaluations to allow for the issuance and tracking of CME/CE certificates. The ISCD may also track aggregate responses to questions in activities and evaluations and use these data to inform the ongoing evaluation and improvement of its educational program. No individual performance data or any other personal information collected for evaluations will be shared with third parties
Attendance Cancellation Policy
All changes in registration must be made in writing to education@iscd.org. Cancellations received 30 days prior to the start of the program are eligible for a full refund minus a $50 administrative fee. Cancellations received less than 30 days prior to the start of the course will not be refunded.
Course Changes or Cancellations
ISCD reserves the right to cancel the program, change dates, how the course is taught, and/or meeting location. If a program is canceled, all program fees will be refunded unless the registered participant elects to transfer to another program. Participants will be notified of any changes within 21 days of the course start date.
-
Register
- Non-Member - $400
- Full Member - $300
- MOC Only - $400
- Community Member - $400
- Professional Member - $400
- More Information
-
Includes Multiple Live Events. The next is on 12/07/2024 at 10:00 AM (EST)
Course Date: December 7 & 8, 2024. The core elements of this course serve to educate healthcare providers about bone densitometry and osteoporosis and to apply standard practices to assure quality performance and interpretation of bone densitometry studies. The course is offered in two parts. The first day sets the standard to perform Quality DXA interpretation. Day 2 focuses on the clinical assessment, prevention, treatment and management of osteoporosis and the essential role that high-quality DXA plays in treatment initiation decisions and monitoring of osteoporotic patients. Individuals may register for either part of the course separately or as a combined program.
Course Date
December 7 & 8, 2024.
Course Description
The core elements of this course serve to educate healthcare providers about bone densitometry and osteoporosis and to apply standard practices to assure quality performance and interpretation of bone densitometry studies. The course is offered in two parts. The first day sets the standard to perform Quality DXA interpretation. Day 2 focuses on the clinical assessment, prevention, treatment and management of osteoporosis and the essential role that high-quality DXA plays in treatment initiation decisions and monitoring of osteoporotic patients. Individuals may register for either part of the course separately or as a combined program.
Target Audience
The course is most appropriate for those involved in the performance and interpretation of DXA scans and those managing patients with osteoporosis. It is open to MDs, PhDs, DOs, Nurse Practitioners, and Physician Assistants. Technologists may attend if they have previously taken the ISCD/IOF Osteoporosis Essentials for Technologists Course.
Learning Objectives
After attending the course, participants should be better prepared to:
- Describe appropriate clinical evaluation of patients at risk for fragility fracture.
- Summarize the value and importance of quality bone densitometry for the diagnosis of osteoporosis, assessment of fracture risk, and monitoring of BMD.
- Recognize errors in DXA analysis and reporting.
- Explain new and emerging guidelines that provide evidence-based information regarding skeletal health.
- Describe the use of current and new pharmacologic therapies for the management and treatment of osteoporosis, including adverse effects.
Method of Participation
This activity will be offered live streamed via Zoom, allowing the learner to watch and ask questions in a live format on December 7 & 8, 2024. Learners will be asked to complete a post-course survey to indicate what they will change in their practice as a result of this activity.
Rates
Cancellation PolicyAttendee Cancellation Policy All changes in registration must be made in writing to education@iscd.org. Cancellations received 30 days prior to the start of the program are eligible for a full refund minus a $50 administrative fee. Cancellations received less than 30 days prior to the start of the course will not be refunded.
Course Changes or Cancellations ISCD reserves the right to cancel the program, change dates and/or meeting location. If a program is canceled, all program fees will be refunded automatically unless the registered participant elects to transfer to another program. Participants will be notified of any changes within 21 days of the course start date.
ACCME Joint Accreditation Statement:
In support of improving patient care, this activity has been planned and implemented by Amedco LLC and ISCD. Amedco LLC is jointly accredited by the Accreditation Council for Continuing Medical Education (ACCME), the Accreditation Council for Pharmacy Education (ACPE), and the American Nurses Credentialing Center (ANCC), to provide continuing education for the healthcare team.

AMA PRA Category 1 Credit(s)™
This activity is under review for accreditation targeting a maximum of 13.00 AMA PRA Category 1 CreditsTM.
Physicians should claim only the credit commensurate with the extent of their participation in the activity.
Deadline to Claim Credit:
Participants will have 90 days from the activity end date to complete the activity survey and claim continuing education credit.
Schedule - December 7th, 2024 - All times Eastern Standard Time (NYC/USA)

Schedule - December 8th, 2024 - All times Eastern Standard Time (NYC/USA)

Dr Neil Binkley MD, CCD
Professor
University of Wisconsin Madison
Dr. Neil Binkley earned his medical degree from the University of Wisconsin and subsequently trained in Internal Medicine at the Marshfield Clinic. After several years in private practice, he returned to the University of Wisconsin and completed a Geriatrics fellowship. He is a Professor in the Divisions of Geriatrics and Endocrinology at the University of Wisconsin School of Medicine and Public Health. He is also Director of the UW Osteoporosis Clinical Research Program and Associate Director of the UW Institute on Aging. He is past President of the International Society for Clinical Densitometry, Associate Editor of Osteoporosis International and a member of the International Osteoporosis Foundation Committee of Scientific Advisors. His current research involves improvement in 25(OH)D measurement, evaluation of approaches to optimize assessment of vitamin D status, evaluation of functional assessment in older adults and the importance of sarcopenia in fracture risk. Dr. Binkley has published over 300 papers, served on expert committees and panels for AACE, ASBMR, IOF and ISCD.

E. Michael Lewiecki, MD, FACP, FACE, CCD
Director
New Mexico Clinical Research & Osteoporosis Center, Inc.
E. Michael Lewiecki, MD, CCD, FACP, FACE, FASBMR, is Director of New Mexico Clinical Research & Osteoporosis Center and Director of Bone Health TeleECHO at University of New Mexico Health Sciences Center in Albuquerque, NM. His is a consultant in osteoporosis and metabolic bone disease, supervisor and interpreter of bone density studies, and an educator with a special interest in the evaluation and treatment of osteoporosis and metabolic bone diseases. He has been principal investigator for many osteoporosis clinical trials and is author of over 300 publications in peer-reviewed medical journals, as well as books, book chapters, and online publications on osteoporosis. Dr. Lewiecki is past-president of the International Society for Clinical Densitometry (ISCD) and current president of the Osteoporosis Foundation of New Mexico (OFNM). He is an editor for Osteoporosis International, and is on the editorial boards of the Journal of Bone and Mineral Research and the Journal of Clinical Densitometry. He has received national and international awards that include the 2021 “Dr. John P. Bilezikian ISCD Global Leadership Award” and the 2021 “Lawrence G. Raisz Memorial Lecture Award” with the Bone Health & Osteoporosis Foundation. Dr. Lewiecki is the program director of the annual Santa Fe Bone Symposium, the flagship activity of the OFNM.

Gillian Wooldridge, DO, CCD
Gillian Wooldridge is a primary care sports medicine physician in Houston, Texas. She completed her undergraduate studies in Kinesiology-Sports Medicine at Rice University in 2009. She received her medical degree from the Texas College of Osteopathic Medicine in 2014, where she was the inaugural recipient of the Sports Medicine Achievement Award. Dr Wooldridge completed her family medicine residency in 2017 and sports medicine fellowship in 2018, with Houston Methodist and Houston Methodist Willowbrook Hospitals respectively.
She currently works for Houston Methodist Orthopedics and Sports Medicine in the Texas Medical Center. She serves as a team physician for Rice University Athletics and volunteers with several organizations in the community including the Chevron Houston Marathon, IronMan Texas and the Houston Livestock Show and Rodeo. Dr Wooldridge is a member of the board of directors for the American Osteopathic Academy of Sports Medicine and is actively involved in the American Medical Society of Sports Medicine as an item writer for the sports medicine in-training exam.
She is a certified clinical densitometrist and works on the Osteoporosis Essentials Course Committee and serves as a co-director of the Bone Health Initiative in the Houston Methodist Orthopedics and Sports Medicine department. She holds a core clinical faculty position with the Texas A&M College of Medicine Engineering & Medicine program and a clinical faculty position the Tillman Fertitta Family College of Medicine.

Statement of Independence
The ISCD maintains a policy on the use of commercial support, which ensures that all educational activities sponsored by the ISCD provide in-depth presentations that are fair, balanced, independent, and scientifically rigorous. ISCD requires faculty, planners, managers, and other individuals and their spouse/life partner who are in a position to control the content of this activity to disclose any real or apparent conflict of interest they may have as related to the content of this activity. All identified conflicts of interest are thoroughly vetted by ISCD for fair balance, scientific objectivity of studies mentioned in the materials or used as the basis for content, and appropriateness of patient care recommendations. Individual disclosures are included in the course material. No Commercial Support has been provided for this activity
Disclaimer
The information and suggestions presented at the courses, seminars and other programs sponsored by ISCD and other collaborating societies are subject to change and therefore should serve only as a foundation for further investigation and study. Any forms presented at our seminars or programs are samples only and are not necessarily authoritative. All information, procedures, and forms contained or used in such seminars or programs should serve only as a guide for use in specific situations.
Disclosure of Unlabeled Use
This educational activity may contain a discussion of published and/or investigational uses of agents that are not indicated by the FDA. ISCD does not recommend the use of any agent outside of the labeled indications. The opinions expressed in this educational activity are those of the faculty and do not necessarily represent the views of any organization associated with this activity. Please refer to the official prescribing information for each product for a discussion of approved indications, contraindications, and warnings.
Privacy & Confidentiality Statement
The ISCD will record the learner’s personal information as provided on evaluations to allow for the issuance and tracking of CME/CE certificates. The ISCD may also track aggregate responses to questions in activities and evaluations and use these data to inform the ongoing evaluation and improvement of its educational program. No individual performance data or any other personal information collected for evaluations will be shared with third parties
Attendance Cancellation Policy
All changes in registration must be made in writing to education@iscd.org. Cancellations received 30 days prior to the start of the program are eligible for a full refund minus a $50 administrative fee. Cancellations received less than 30 days prior to the start of the course will not be refunded.
Course Changes or Cancellations
ISCD reserves the right to cancel the program, change dates, how the course is taught, and/or meeting location. If a program is canceled, all program fees will be refunded unless the registered participant elects to transfer to another program. Participants will be notified of any changes within 21 days of the course start date.
-
Register
- Non-Member - $800
- Full Member - $500
- MOC Only - $800
- Community Member - $800
- Professional Member - $800
- More Information
-
Includes Multiple Live Events. The next is on 10/20/2024 at 10:00 AM (EDT)
Course Date: October 20, 2024. This course focuses on increasing the knowledge and competence of clinicians and other healthcare professionals to help them successfully use DXA body composition analysis in the management of obesity, geriatric sarcopenia and other low muscle mass states, general health and pediatric skeletal disease. The inclusion of case studies will reinforce the knowledge gained and allow for application in the diagnosis, treatment and therapies of patients presenting with low bone mineral density.
Course Date
October 20, 2024, 10am-2pm ET
Course Description
This course focuses on increasing the knowledge and competence of clinicians and other healthcare professionals to help them successfully use DXA body composition analysis in the management of obesity, geriatric sarcopenia and other low muscle mass states, general health and pediatric skeletal disease. The inclusion of case studies will reinforce the knowledge gained and allow for application in the diagnosis, treatment and therapies of patients presenting with low bone mineral density.
Target Audience
The content is designed for healthcare providers responsible for reading and interpreting DXA scans, including specialists and generalists working in family medicine, general and internal medicine, rheumatology, endocrinology, obstetrics/gynecology, radiology, physical and occupational therapy, and research, as well as technologists.
Learning Objectives
After attending the course, participants should be better prepared to:
- Identify proper DXA technology for body composition analysis of fat, lean muscle and bone mass.
- List other methods of body composition analysis used in the clinical setting.
- Differentiate reference values and indices.
- Identify limitations of reference values and indices.
- Summarize scan acquisition for adult and pediatric patients.
Method of Participation
This activity will be offered through zoom, allowing the learner to watch and ask questions in a live format on October 20, 2024. At the end of the activity, learners will be asked to evaluate the activity online and then claim credit and print certificates. Participants will have 90 days from the activity end date to complete the activity survey and claim continuing education credit.
Rates
Cancellation Policy
Attendee Cancellation Policy All changes in registration must be made in writing to education@iscd.org. Cancellations received 30 days prior to the start of the program are eligible for a full refund minus a $50 administrative fee. Cancellations received less than 30 days prior to the start of the course will not be refunded.
Course Changes or Cancellations ISCD reserves the right to cancel the program, change dates and/or meeting location. If a program is canceled, all program fees will be refunded automatically unless the registered participant elects to transfer to another program. Participants will be notified of any changes within 21 days of the course start date.
ACCME Joint Accreditation Statement

In support of improving patient care, this activity has been planned and implemented by Amedco LLC and ISCD. Amedco LLC is jointly accredited by the Accreditation Council for Continuing Medical Education (ACCME), the Accreditation Council for Pharmacy Education (ACPE), and the American Nurses Credentialing Center (ANCC), to provide continuing education for the healthcare team.
AMA PRA Category 1 Credit(s)™
This activity is currently under review for accreditation targeting 3.75 AMA PRA Category 1 Credit(s)TM.
ASRT Credit Designation:
This activity has been approved by ASRT for 4.5 Category A continuing education credits.
Deadline to Claim Credit
Participants will have 90 days from the activity end date to complete the activity survey and claim continuing education credit.
Schedule - All times eastern (NYC/USA)

Greg Yardley
Sr. Medical Radiology Technologist and Researcher
NASA Bone Mineral Laboratory
Greg Yardley works as a SR. Medical Radiology Technologist and researcher in the NASA Bone Mineral Laboratory and NASA/JSC medical clinic for KBR in the government solutions division. His job requirements are performing and analyzing, Dual Energy x-ray absorptiometry (DXA) exams on both retired and active Astronauts. He also extracts, records, aids in bone density interpretation and distributes bone data from Dual Energy xray absorptiometry (DXA), Computed Tomography (CT), Magnetic resonance imaging devises. Greg also performs diagnostic medical x-rays for the NASA/JSC Flight medicine and occupational health clinic.
Greg began his healthcare career as an Emergency Medical Technician (EMT). He then furthered his medical education and received his certification as a registered radiology technologist. Upon completion of his Radiological Science courses. Greg has worked as a Radiology technologist in a Trauma level 1 Hospital, Level III hospital, urgent care, and multi-specialty clinic. At the time of his graduation Greg had also received extensive training not only in X-ray but also DXA, CT, and Ultrasound. In 2011 just previous to his current job Greg got a job as a Bone Density Technologist at a multi-specialty medical clinic where he performed and analyzed over 3000 DXA scans a year on every type of patient imaginable while also running the day-to-day operations of the Osteoporosis center.

Michelle Swainson, PhD
Lecturer in Physiology
Lancaster Medical School, Lancaster University
Michelle Swainson, PhD is a UK-based exercise physiologist with an honours degree in Sport and Exercise Science (Kinesiology), a master’s degree in Exercise Physiology, and a PhD focusing on fitness, body composition and cardiovascular disease risk in healthy adults. Michelle is a Lecturer in Physiology at Lancaster Medical School, Lancaster University, where her teaching includes clinical exercise physiology, public health challenges including obesity and ageing, exercise medicine, and optimising health outcomes through programme evaluation and interventions. She is the DXA lead for the new Health Innovation Imaging Suite, where her current research is focused on the assessment of visceral adipose tissue using DXA in both general and athletic populations, with specific interest on its use to predict cardiometabolic disease risk factors. Michelle is a member of the ISCD Body Composition Education Committee, a British Association for the Sport and Exercise Sciences (BASES) Accredited Sport and Exercise Scientist, a committee member of the BASES Education and Teaching Special Interest Group, and the North West Network Representative for the UK Association for the Study of Obesity.

Statement of independence
The ISCD maintains a policy on the use of commercial support, which ensures that all educational activities sponsored by the ISCD provide in-depth presentations that are fair, balanced, independent, and scientifically rigorous. ISCD requires faculty, planners, managers, and other individuals and their spouse/life partner who are in a position to control the content of this activity to disclose any real or apparent conflict of interest they may have as related to the content of this activity. All identified conflicts of interest are thoroughly vetted by ISCD for fair balance, scientific objectivity of studies mentioned in the materials or used as the basis for content, and appropriateness of patient care recommendations. Individual disclosures are included in the course material. No Commercial Support has been provided for this activity
Disclaimer
The information and suggestions presented at the courses, seminars and other programs sponsored by ISCD and other collaborating societies are subject to change and therefore should serve only as a foundation for further investigation and study. Any forms presented at our seminars or programs are samples only and are not necessarily authoritative. All information, procedures, and forms contained or used in such seminars or programs should serve only as a guide for use in specific situations.
Disclosure of Unlabeled Use
This educational activity may contain a discussion of published and/or investigational uses of agents that are not indicated by the FDA. ISCD does not recommend the use of any agent outside of the labeled indications. The opinions expressed in this educational activity are those of the faculty and do not necessarily represent the views of any organization associated with this activity. Please refer to the official prescribing information for each product for a discussion of approved indications, contraindications, and warnings.
Privacy & Confidentiality Statement
The ISCD will record the learner’s personal information as provided on evaluations to allow for the issuance and tracking of CME/CE certificates. The ISCD may also track aggregate responses to questions in activities and evaluations and use these data to inform the ongoing evaluation and improvement of its educational program. No individual performance data or any other personal information collected for evaluations will be shared with third parties
Attendance Cancellation Policy
All changes in registration must be made in writing to education@iscd.org. Cancellations received 30 days prior to the start of the program are eligible for a full refund minus a $50 administrative fee. Cancellations received less than 30 days prior to the start of the course will not be refunded.
Course Changes or Cancellations
ISCD reserves the right to cancel the program, change dates, how the course is taught, and/or meeting location. If a program is canceled, all program fees will be refunded unless the registered participant elects to transfer to another program. Participants will be notified of any changes within 21 days of the course start date.
-
Register
- Non-Member - $200
- Full Member - $100
- MOC Only - $200
- Community Member - $200
- Professional Member - $200
- More Information
-
Includes Multiple Live Events. The next is on 10/19/2024 at 10:00 AM (EDT)
Course Date: October 19, 2024. The Pediatric Bone Densitometry Course provides the most current research and best-practice techniques for obtaining and interpreting bone mineral density (BMD) measurements in children and adolescents. This educational program is designed to improve learner competence in the area of pediatric densitometry. Learners will have the opportunity to take the knowledge gained from this educational program and properly/consistently apply it to their professional practices.
Course Date
October 19, 2024
Course Description
The Pediatric Bone Densitometry Course provides current research and best-practice techniques for the acquisition and interpretation of bone mineral density (BMD) in children and adolescents. This educational program is designed to improve learner competence in pediatric bone densitometry. By completing this course, attendees will gain conceptual and practical knowledge which can be applied in their professional practices.
Target Audience
The content is designed for healthcare providers with an interest in skeletal health assessment, including specialists, generalists and technologists working in family medicine, general and internal medicine, endocrinology, rheumatology, obstetrics/gynecology, radiology, pediatrics, physical and occupational therapy, nuclear medicine and research.
Learning Objectives
After attending the course, participants should be better prepared to:
1. Recognize considerations and concerns with skeletal assessment in children and adolescents with a variety of medical conditions.
2. Identify the most appropriate and reproducible sites for densitometry in children and adolescents.
3. Evaluate technical aspects of bone assessment in children and adolescents.
4. Describe the uses of measurement tools for acquiring bone densitometry assessments by DXA and other modalities.
5. Summarize the elements to be included in a pediatric DXA report.
Method of Participation
This activity will be offered through Zoom, allowing the learner to watch and ask questions in a live format on October 19, 2024. At the end of the activity, learners will be asked to evaluate the activity online and then claim credit and print certificates. Participants will have 90 days from the activity end date to complete the activity survey and claim continuing education credit.
Rates
Cancellation Policy
Attendee Cancellation Policy All changes in registration must be made in writing to education@iscd.org. Cancellations received 30 days prior to the start of the program are eligible for a full refund minus a $50 administrative fee. Cancellations received less than 30 days prior to the start of the course will not be refunded.
Course Changes or Cancellations ISCD reserves the right to cancel the program, change dates and/or meeting location. If a program is canceled, all program fees will be refunded automatically unless the registered participant elects to transfer to another program. Participants will be notified of any changes within 21 days of the course start date.
ACCME Joint Accreditation Statement:

In support of improving patient care, this activity has been planned and implemented by Amedco LLC and ISCD. Amedco LLC is jointly accredited by the Accreditation Council for Continuing Medical Education (ACCME), the Accreditation Council for Pharmacy Education (ACPE), and the American Nurses Credentialing Center (ANCC), to provide continuing education for the healthcare team.
AMA PRA Category 1 Credit(s)™
This activity is currently under review for accreditation targeting 3.75 AMA PRA Category 1 Credit(s)TM.
ASRT Credit Designation:
Successful completion of this activity qualifies for 4.5 Category A continuing education credits.
Deadline to Claim Credit:
Participants will have 90 days from the activity end date to complete the activity survey and claim continuing education credit.
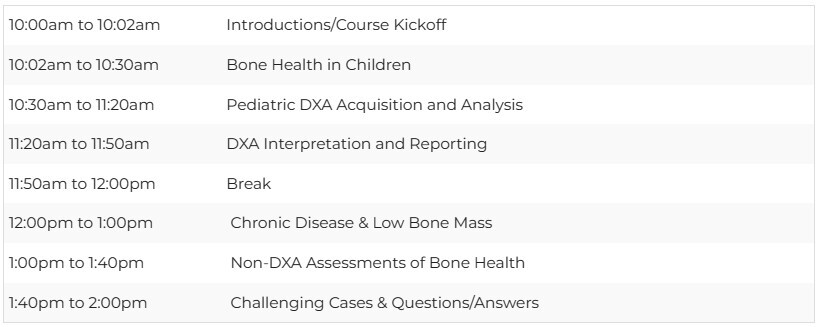
Schedule - October 19, 2024 - All times eastern (NYC/USA)

Kyla Kent, BA, CBDT
Technical Director
SAMBA Lab, Stanford University School of Medicine
Kyla Kent, BA, CBDT, Technical Director of the SAMBA (Stanford Assessments of Muscle and Bone across the Ages) Lab, has been with Stanford University School of Medicine and the Palo Alto Veterans Affairs Health Care system since 1991 in the Stanford Prevention Research Center, the Musculo-Skeletal Research Lab and most recently the SAMBA Lab. Responsibilities include development and technical assessment of research protocols, clinical facility management, supervisor for clinical staff, performance of DXA, HRpQCT, Biodex and other measures related to research conducted in the areas of bone metabolism, structural assessment, body composition, exercise, sleep and healthy aging. Ms. Kent is a consultant in study design and instructor in bone density related imaging modalities for the Stanford Children’s Hospital, Palo Alto VA Hospital and Stanford Hospital/Stanford School of Medicine and internationally for bone mass measurement technique and analysis in growing skeletons. Currently acting in the position of Technical Director for the SAMBA Lab and as consultant developing a program for Quality DXA across all of Stanford’s clinical networks. Ms. Kent is an active member of the International Society for Clinical Densitometry (ISCD. She also has acted as the Director, clinical instructor and lecturer for a State certified training program for DXA.

John J. Carey, MBBChBAO, MS, CCD, FRCPI
Physician in Rheumatology, Osteology, and Medicine / Professor in Medicine
Galway University Hospitals / University of Galway
Prof. John J. Carey is a consultant physician in rheumatology and medicine, and clinical director of DXA, FLS programme and osteoporosis and metabolic bone disease at Galway University Hospitals, Ireland, and a Professor in Medicine at The National University of Ireland, Galway. He completed medical school in Ireland, and his post-graduate training in medicine, rheumatology and clinical research in the United States of America. He is a past-president of The International Society for Clinical Densitometry, current president of The Irish DXA Society and a member of The Committee for Scientific Affairs for the International Osteoporosis Foundation. He is faculty for the ISCD OE, Pediatric, VFR and Body Composition courses. His research interests focus on evidence synthesis and reality-based medicine, and optimizing the use of electronic health information, diagnostic testing and evidence in clinical practice.

Statement of Independence
The ISCD maintains a policy on the use of commercial support, which ensures that all educational activities sponsored by the ISCD provide in-depth presentations that are fair, balanced, independent, and scientifically rigorous. ISCD requires faculty, planners, managers, and other individuals and their spouse/life partner who are in a position to control the content of this activity to disclose any real or apparent conflict of interest they may have as related to the content of this activity. All identified conflicts of interest are thoroughly vetted by ISCD for fair balance, scientific objectivity of studies mentioned in the materials or used as the basis for content, and appropriateness of patient care recommendations. Individual disclosures are included in the course material. No Commercial Support has been provided for this activity
Disclaimer
The information and suggestions presented at the courses, seminars and other programs sponsored by ISCD and other collaborating societies are subject to change and therefore should serve only as a foundation for further investigation and study. Any forms presented at our seminars or programs are samples only and are not necessarily authoritative. All information, procedures, and forms contained or used in such seminars or programs should serve only as a guide for use in specific situations.
Disclosure of Unlabeled Use
This educational activity may contain a discussion of published and/or investigational uses of agents that are not indicated by the FDA. ISCD does not recommend the use of any agent outside of the labeled indications. The opinions expressed in this educational activity are those of the faculty and do not necessarily represent the views of any organization associated with this activity. Please refer to the official prescribing information for each product for a discussion of approved indications, contraindications, and warnings.
Privacy & Confidentiality Statement
The ISCD will record the learner’s personal information as provided on evaluations to allow for the issuance and tracking of CME/CE certificates. The ISCD may also track aggregate responses to questions in activities and evaluations and use these data to inform the ongoing evaluation and improvement of its educational program. No individual performance data or any other personal information collected for evaluations will be shared with third parties
Attendance Cancellation Policy
All changes in registration must be made in writing to education@iscd.org. Cancellations received 30 days prior to the start of the program are eligible for a full refund minus a $50 administrative fee. Cancellations received less than 30 days prior to the start of the course will not be refunded.
Course Changes or Cancellations
ISCD reserves the right to cancel the program, change dates, how the course is taught, and/or meeting location. If a program is canceled, all program fees will be refunded unless the registered participant elects to transfer to another program. Participants will be notified of any changes within 21 days of the course start date.
-
Register
- Non-Member - $200
- Full Member - $100
- MOC Only - $200
- Community Member - $200
- Professional Member - $200
- More Information
-
Register
-
Includes Multiple Live Events. The next is on 10/02/2024 at 10:00 AM (EDT)
Course Date: October 2, 2024. This advanced densitometry course focuses on how vertebral fracture imaging advances the diagnosis and treatment of patients at risk of osteoporotic fractures. It emphasizes the importance of vertebral fractures in the clinical management of osteoporosis, including recognition of lateral spine images of a normal spine, vertebral fractures, and other findings that may be confused with vertebral fractures. Fracture recognition, analysis, and best-practice patient positioning are detailed to ensure ideal image acquisition, interpretation, and reporting.
Course Date
October 2, 2024.
Course Description
This advanced densitometry course focuses on how vertebral fracture imaging advances the diagnosis and treatment of patients at risk of osteoporotic fractures. It emphasizes the importance of vertebral fractures in the clinical management of osteoporosis, including recognition of lateral spine images of a normal spine, vertebral fractures, and other findings that may be confused with vertebral fractures. Fracture recognition, analysis, and best-practice patient positioning are detailed to ensure ideal image acquisition, interpretation, and reporting. It is recommended, but not required, that registrants attend the Osteoporosis Essentials/Quality Bone Densitometry Course (Clinician or Technologist) prior to attending the VFR course
Target Audience
The content is designed for healthcare providers responsible for reading and interpreting DXA scans, including specialists and generalists working in family medicine, general and internal medicine, rheumatology, endocrinology, obstetrics/gynecology, radiology, nuclear medicine, physical and occupational therapy, and research. The course is also appropriate for technologists.
Learning Objectives
After attending the course, participants should be better prepared to:
1. Summarize best-practice methods utilized to recognize vertebral fractures on lateral spine images.
2. Describe non-fracture abnormalities that may appear on a spine image.
3. Identify characteristics of high-quality densitometric lateral spine images.
4. Identify the appropriate complementary technologies to complete vertebral fracture assessment.
5. Cite indications for VFA as stated in the 2015 ISCD Official Positions.
Method of Participation
This activity will be offered through zoom, allowing the learner to watch and ask questions in a live format on October 2, 2024. At the end of the activity, learners will be asked to evaluate the activity online and then claim credit and print certificates. Participants will have 90 days from the activity end date to complete the activity survey and claim continuing education credit.
Rates
Cancellation Policy
Attendee Cancellation Policy All changes in registration must be made in writing to education@iscd.org. Cancellations received 30 days prior to the start of the program are eligible for a full refund minus a $50 administrative fee. Cancellations received less than 30 days prior to the start of the course will not be refunded.
Course Changes or Cancellations ISCD reserves the right to cancel the program, change dates and/or meeting location. If a program is canceled, all program fees will be refunded automatically unless the registered participant elects to transfer to another program. Participants will be notified of any changes within 21 days of the course start date.
ACCME Joint Accreditation Statement:

In support of improving patient care, this activity has been planned and implemented by Amedco LLC and ISCD. Amedco LLC is jointly accredited by the Accreditation Council for Continuing Medical Education (ACCME), the Accreditation Council for Pharmacy Education (ACPE), and the American Nurses Credentialing Center (ANCC), to provide continuing education for the healthcare team.
AMA PRA Category 1 Credit(s)™
Amedco LLC designates this live activity for a maximum of 3.75 AMA PRA Category 1 Credit(s)TM.
Physicians should claim only the credit commensurate with the extent of their participation in the activity.
ASRT Credit Designation
This activity has been approved by ASRT for 4.5 Category A continuing education credits.
Deadline to Claim Credit
Participants will have 90 days from the activity end date to complete the activity survey and claim continuing education credit.
Schedule - All times eastern (NYC/USA)

Dr. Harold Rosen, MD, CCD
Director of the Osteoporosis Prevention and Treatment Center
Beth Israel Deaconess Medical Center
Dr. Harold Rosen did fellowships in Endocrinology and Geriatrics at Beth Israel Hospital in Boston. He spent years doing clinical research in osteoporosis, when he investigated the relationship between vitamin K and bone, thyroid hormone-induced bone loss, and the role of biochemical markers of bone turnover. In 1998 he became the Director of the Osteoporosis Prevention and Treatment Center at Beth Israel Deaconess Medical Center in Boston. In this capacity he runs bone densitometry, sees patients in consultation for osteoporosis and metabolic bone disease, and does research on optimizing performance and reporting of bone densitometry. He has been active in his professional society, the ISCD (International Society for Clinical Densitometry) where he served on the Board of Directors from 2010-2013. Dr. Rosen is the recipient of the ISCD Clinician of the year Award in 2010, and the recipient of the Paul Miller award for service to the ISCD in 2017.

John Schousboe, MD, PhD, CCD
Director, Park Nicollet Bone Densitometry Center; Consultant Rheumatologist, Park Nicollet Clinic; Research Investigator, HealthPartners Institute
Dr. Schousboe is a rheumatologist and health services researcher who directs the bone densitometry center of Park Nicollet Clinic, a large integrated health care delivery system in Minneapolis, MN. He is a Past President of the International Society for Clinical Densitometry (ISCD) and a Fellow of the American Society for Bone and Mineral Research (ASBMR). During a 16-year research career he has been an author on 200 peer-reviewed publications, the far majority of which have been in the field of diagnosis and management of osteoporosis. His contributions to science include development of a cost-effectiveness model that was used to conduct studies of the cost-effectiveness of bone densitometry in men and subgroups of older women and men defined by prevalent vertebral fracture, bone turnover marker levels, and bone density level; studies of the accuracy, prevalence, and clinical impact of vertebral fracture recognition on bone density lateral spine images; studies of medication adherence to osteoporosis therapies; studies of the identification and clinical significance of abdominal aortic calcification on lateral bone density spine images; and predictors of health care utilization and costs using Medicare Claims data.

Statement of Independence
The ISCD maintains a policy on the use of commercial support, which ensures that all educational activities sponsored by the ISCD provide in-depth presentations that are fair, balanced, independent, and scientifically rigorous. ISCD requires faculty, planners, managers, and other individuals and their spouse/life partner who are in a position to control the content of this activity to disclose any real or apparent conflict of interest they may have as related to the content of this activity. All identified conflicts of interest are thoroughly vetted by ISCD for fair balance, scientific objectivity of studies mentioned in the materials or used as the basis for content, and appropriateness of patient care recommendations. Individual disclosures are included in the course material. No Commercial Support has been provided for this activity
Disclaimer
The information and suggestions presented at the courses, seminars and other programs sponsored by ISCD and other collaborating societies are subject to change and therefore should serve only as a foundation for further investigation and study. Any forms presented at our seminars or programs are samples only and are not necessarily authoritative. All information, procedures, and forms contained or used in such seminars or programs should serve only as a guide for use in specific situations.
Disclosure of Unlabeled Use
This educational activity may contain a discussion of published and/or investigational uses of agents that are not indicated by the FDA. ISCD does not recommend the use of any agent outside of the labeled indications. The opinions expressed in this educational activity are those of the faculty and do not necessarily represent the views of any organization associated with this activity. Please refer to the official prescribing information for each product for a discussion of approved indications, contraindications, and warnings.
Privacy & Confidentiality Statement
The ISCD will record the learner’s personal information as provided on evaluations to allow for the issuance and tracking of CME/CE certificates. The ISCD may also track aggregate responses to questions in activities and evaluations and use these data to inform the ongoing evaluation and improvement of its educational program. No individual performance data or any other personal information collected for evaluations will be shared with third parties
Attendance Cancellation Policy
All changes in registration must be made in writing to education@iscd.org. Cancellations received 30 days prior to the start of the program are eligible for a full refund minus a $50 administrative fee. Cancellations received less than 30 days prior to the start of the course will not be refunded.
Course Changes or Cancellations
ISCD reserves the right to cancel the program, change dates, how the course is taught, and/or meeting location. If a program is canceled, all program fees will be refunded unless the registered participant elects to transfer to another program. Participants will be notified of any changes within 21 days of the course start date.
-
Register
- Non-Member - $200
- Full Member - $100
- MOC Only - $200
- Community Member - $200
- Professional Member - $200
- More Information
-
Register
-
Includes Multiple Live Events. The next is on 09/21/2024 at 10:00 AM (EDT)
Course Date: September 21 & 22, 2024. The core elements of this program focus on performing quality DXA scans, dissecting the data and applying this knowledge to a technologist’s role in patient care. This course, which focuses on quality DXA, is innovative, interactive and informative. The course emphasizes technical excellence in the performance, analysis and troubleshooting of bone densitometry scans. It provides standard practices and applications to raise the quality of DXA scan procedures and their impact on patient care. The program is designed to enhance awareness of the technical expertise involved in performing bone densitometry and applying protocols to address the everyday challenges. Attendees with come away with a strong sense of the considerable impact that a technologist plays in the delivery of quality DXA, and how that influences patient management.
Course Date
September 21 & 22, 2024.
Course Description
The core elements of this program focus on performing quality DXA scans, dissecting the data and applying this knowledge to a technologist’s role in patient care. This course, which focuses on quality DXA, is innovative, interactive and informative. The course emphasizes technical excellence in the performance, analysis and troubleshooting of bone densitometry scans. It provides standard practices and applications to raise the quality of DXA scan procedures and their impact on patient care. The program is designed to enhance awareness of the technical expertise involved in performing bone densitometry and applying protocols to address the everyday challenges. Attendees with come away with a strong sense of the considerable impact that a technologist plays in the delivery of quality DXA, and how that influences patient management.
Target Audience
The course is most appropriate for those involved in the performance of DXA scans and open to any health professional performing these scans.
Learning Objectives
After attending the course, participants should be better prepared to:
- Design and implement quality control and assurance practices.
- Perform a precision assessment and appreciate the requirement for clinical care.
- Identify the anatomy relevant to clinical DXA scanning.
- Implement consistent and appropriate technical acquisition and analysis techniques.
- Perform a fracture risk assessment using FRAX.
- Provide DXA interpreters sufficient information for a high-quality DXA report.
- Discuss supplemental software and their application.
- Demonstrate how to access features within the DXA software.
- Understand how specific medical conditions and medications affect bone density.
- Critically evaluate DXA scans to identify acquisition and analysis errors and implement resolution methods.
- Recognize how technical errors impact clinical interpretation and patient care.
Method of Participation
This activity will be offered through zoom, allowing the learner to watch and ask questions in a live format on September 21 & 22, 2024. Learners will be asked to complete a post-course survey to indicate what they will change in their practice as a result of this activity.
Registration Fee
Cancellation Policy
Attendee Cancellation Policy All changes in registration must be made in writing to education@iscd.org. Cancellations received 30 days prior to the start of the program are eligible for a full refund minus a $50 administrative fee. Cancellations received less than 30 days prior to the start of the course will not be refunded.
Course Changes or Cancellations ISCD reserves the right to cancel the program, change dates and/or meeting location. If a program is canceled, all program fees will be refunded automatically unless the registered participant elects to transfer to another program. Participants will be notified of any changes within 21 days of the course start date.
ASRT Credit Designation
This activity has been approved by ASRT for 13.0 Category A continuing education credits.
ASRT Credit Notice
This course is a renewal. If you have taken the course since June 2022, you will not be able to claim ASRT again for this learning activity. If you have taken prior to June 2022, you can take this course again for credit.
Deadline to Claim Credit
Participants will have 90 days from the activity end date to complete the activity survey and claim continuing education credit.
Schedule - Day 1 - All times Eastern (NYC/USA)
Schedule - Day 2 - All times Eastern (NYC/USA)

Anita Colquhoun, RTNM, MRT(N), CDT
Charge Technologist
Women's College Hospital
Anita Colquhoun M.R.T.(N), CDT is the charge technologist in the Centre for Osteoporosis & Bone Health at the Women’s College Hospital in Toronto. Anita is a Nuclear Medical Technologist specialized in Bone Mineral Density (BMD). She has been working in the field of BMD since the early 1990s. Anita is an active member ISCD Technologist Faculty since 2000. She has served 2 terms on the Governing Board of the ISCD and is currently serving her third term as the Executive Secretary to the Board. As well as Chairing then Annual Meeting Committee from 2012-2016 Anita served and is still serving on several other committees within the ISCD. She has participated in a variety of research studies and has co-authored abstracts and papers. Anita has also co-written an on-line beginner BMD course as well as developed a competency profile and best practise in BMD for the Ontario Association of Medical Radiation Sciences.

Lawrence Jankowski, CBDT
Chief DXA Technologist/Research Coordinator
Illinois Bone and Joint Institute, LLC
Lawrence Jankowski is the chief DXA technologist and a research coordinator for Illinois Bone and Joint Institute, a multi-disciplinary 100+ physician practice with over 20 locations within Chicago and surrounding suburbs, specializing in orthopedic and rheumatologic health and sports performance and fitness. He started clinical imaging as a nuclear medicine technologist in 1978, performing both single- and dual- photon absorptiometry in beginning in 1987, and switched to DXA in 1990. He is also registered in radiology (limited), and is certified by the International Society for Clinical Densitometry (ISCD) first as a CDT in 1996, and then CBDT in 2001.
In addition to his clinical and research duties, he is a technical instructor for the ISCD bone density courses, serves on the editorial board for the Journal of Clinical Densitometry, chairman of the Facility Accreditation Program, He has presented numerous posters on bone densitometry at scientific meetings, including the ASBMR, AAOS, NOF, as well as ISCD. Most recently, he served as task force chair for the 2019 Position Development Conference on cross-calibration and LSC determination in multiple scanner environments.
Greg Yardley
Sr. Medical Radiology Technologist and Researcher
NASA Bone Mineral Laboratory
Greg Yardley works as a SR. Medical Radiology Technologist and researcher in the NASA Bone Mineral Laboratory and NASA/JSC medical clinic for KBR in the government solutions division. His job requirements are performing and analyzing, Dual Energy x-ray absorptiometry (DXA) exams on both retired and active Astronauts. He also extracts, records, aids in bone density interpretation and distributes bone data from Dual Energy xray absorptiometry (DXA), Computed Tomography (CT), Magnetic resonance imaging devises. Greg also performs diagnostic medical x-rays for the NASA/JSC Flight medicine and occupational health clinic.
Greg began his healthcare career as an Emergency Medical Technician (EMT). He then furthered his medical education and received his certification as a registered radiology technologist. Upon completion of his Radiological Science courses. Greg has worked as a Radiology technologist in a Trauma level 1 Hospital, Level III hospital, urgent care, and multi-specialty clinic. At the time of his graduation Greg had also received extensive training not only in X-ray but also DXA, CT, and Ultrasound. In 2011 just previous to his current job Greg got a job as a Bone Density Technologist at a multi-specialty medical clinic where he performed and analyzed over 3000 DXA scans a year on every type of patient imaginable while also running the day-to-day operations of the Osteoporosis center.

Statement of Independence
The ISCD maintains a policy on the use of commercial support, which ensures that all educational activities sponsored by the ISCD provide in-depth presentations that are fair, balanced, independent, and scientifically rigorous. ISCD requires faculty, planners, managers, and other individuals and their spouse/life partner who are in a position to control the content of this activity to disclose any real or apparent conflict of interest they may have as related to the content of this activity. All identified conflicts of interest are thoroughly vetted by ISCD for fair balance, scientific objectivity of studies mentioned in the materials or used as the basis for content, and appropriateness of patient care recommendations. Individual disclosures are included in the course material. No Commercial Support has been provided for this activity
Disclaimer
The information and suggestions presented at the courses, seminars and other programs sponsored by ISCD and other collaborating societies are subject to change and therefore should serve only as a foundation for further investigation and study. Any forms presented at our seminars or programs are samples only and are not necessarily authoritative. All information, procedures, and forms contained or used in such seminars or programs should serve only as a guide for use in specific situations.
Disclosure of Unlabeled Use
This educational activity may contain a discussion of published and/or investigational uses of agents that are not indicated by the FDA. ISCD does not recommend the use of any agent outside of the labeled indications. The opinions expressed in this educational activity are those of the faculty and do not necessarily represent the views of any organization associated with this activity. Please refer to the official prescribing information for each product for a discussion of approved indications, contraindications, and warnings.
Privacy & Confidentiality Statement
The ISCD will record the learner’s personal information as provided on evaluations to allow for the issuance and tracking of CME/CE certificates. The ISCD may also track aggregate responses to questions in activities and evaluations and use these data to inform the ongoing evaluation and improvement of its educational program. No individual performance data or any other personal information collected for evaluations will be shared with third parties
Attendance Cancellation Policy
All changes in registration must be made in writing to education@iscd.org. Cancellations received 30 days prior to the start of the program are eligible for a full refund minus a $50 administrative fee. Cancellations received less than 30 days prior to the start of the course will not be refunded.
Course Changes or Cancellations
ISCD reserves the right to cancel the program, change dates, how the course is taught, and/or meeting location. If a program is canceled, all program fees will be refunded unless the registered participant elects to transfer to another program. Participants will be notified of any changes within 21 days of the course start date.
-
Register
- Non-Member - $400
- Full Member - $300
- MOC Only - $400
- Community Member - $400
- Professional Member - $400
- More Information
-
Includes Multiple Live Events. The next is on 09/21/2024 at 10:00 AM (EDT)
Course Date: September 21 & 22, 2024. The core elements of this course serve to educate healthcare providers about bone densitometry and osteoporosis and to apply standard practices to assure quality performance and interpretation of bone densitometry studies. The course is offered in two parts. The first day sets the standard to perform Quality DXA interpretation. Day 2 focuses on the clinical assessment, prevention, treatment and management of osteoporosis and the essential role that high-quality DXA plays in treatment initiation decisions and monitoring of osteoporotic patients. Individuals may register for either part of the course separately or as a combined program.
Course Date
September 21 & 22, 2024.
Course Description
The core elements of this course serve to educate healthcare providers about bone densitometry and osteoporosis and to apply standard practices to assure quality performance and interpretation of bone densitometry studies. The course is offered in two parts. The first day sets the standard to perform Quality DXA interpretation. Day 2 focuses on the clinical assessment, prevention, treatment and management of osteoporosis and the essential role that high-quality DXA plays in treatment initiation decisions and monitoring of osteoporotic patients. Individuals may register for either part of the course separately or as a combined program.
Target Audience
The course is most appropriate for those involved in the performance and interpretation of DXA scans and those managing patients with osteoporosis. It is open to MDs, PhDs, DOs, Nurse Practitioners, and Physician Assistants. Technologists may attend if they have previously taken the ISCD/IOF Osteoporosis Essentials for Technologists Course.
Learning Objectives
After attending the course, participants should be better prepared to:
- Describe appropriate clinical evaluation of patients at risk for fragility fracture.
- Summarize the value and importance of quality bone densitometry for the diagnosis of osteoporosis, assessment of fracture risk, and monitoring of BMD.
- Recognize errors in DXA analysis and reporting.
- Explain new and emerging guidelines that provide evidence-based information regarding skeletal health.
- Describe the use of current and new pharmacologic therapies for the management and treatment of osteoporosis, including adverse effects.
Method of Participation
This activity will be offered live streamed via Zoom, allowing the learner to watch and ask questions in a live format on September 21 & 22, 2024. Learners will be asked to complete a post-course survey to indicate what they will change in their practice as a result of this activity.
Rates
Cancellation PolicyAttendee Cancellation Policy All changes in registration must be made in writing to education@iscd.org. Cancellations received 30 days prior to the start of the program are eligible for a full refund minus a $50 administrative fee. Cancellations received less than 30 days prior to the start of the course will not be refunded.
Course Changes or Cancellations ISCD reserves the right to cancel the program, change dates and/or meeting location. If a program is canceled, all program fees will be refunded automatically unless the registered participant elects to transfer to another program. Participants will be notified of any changes within 21 days of the course start date.
ACCME Joint Accreditation Statement
In support of improving patient care, this activity has been planned and implemented by Amedco LLC and ISCD. Amedco LLC is jointly accredited by the Accreditation Council for Continuing Medical Education (ACCME), the Accreditation Council for Pharmacy Education (ACPE), and the American Nurses Credentialing Center (ANCC), to provide continuing education for the healthcare team.

AMA PRA Category 1 Credit(s)™
This activity is under review for accreditation targeting a maximum of 13.00 AMA PRA Category 1 CreditsTM.
Physicians should claim only the credit commensurate with the extent of their participation in the activity.
Deadline to Claim Credit
Participants will have 90 days from the activity end date to complete the activity survey and claim continuing education credit.
Schedule - September 21st, 2024 - All times Eastern Standard Time (NYC/USA)

Schedule - September 22nd, 2024 - All times Eastern Standard Time (NYC/USA)

Dr. Sanford Baim, MD, FACR, CCD
Professor of Medicine, Director of Bone Metabolic Disease, Division of Endocrinology and Metabolism
RUSH University Medical Center
Dr. Sanford Baim is Professor of Medicine, Director of Bone Metabolic Disease, Division of Endocrinology and Metabolism at RUSH University Medical Center. He is past president of the International Society for Clinical Densitometry, has published numerous articles, participated in the development of an interactive web-based teaching tool for bone densitometry and is presently engaged in osteoporosis clinical outcomes research. Additionally, he has served on the leadership boards of numerous conferences and steering committees tasked with setting official positions on the diagnoses and treatment of metabolic bone diseases, such as osteoporosis. Dr. Baim is currently developing a comprehensive, multidisciplinary, integrative strategic plan for the prevention of osteoporosis throughout the Rush University medical system.

E. Michael Lewiecki, MD, FACP, FACE, CCD
Director
New Mexico Clinical Research & Osteoporosis Center, Inc.
E. Michael Lewiecki, MD, CCD, FACP, FACE, FASBMR, is Director of New Mexico Clinical Research & Osteoporosis Center and Director of Bone Health TeleECHO at University of New Mexico Health Sciences Center in Albuquerque, NM. His is a consultant in osteoporosis and metabolic bone disease, supervisor and interpreter of bone density studies, and an educator with a special interest in the evaluation and treatment of osteoporosis and metabolic bone diseases. He has been principal investigator for many osteoporosis clinical trials and is author of over 300 publications in peer-reviewed medical journals, as well as books, book chapters, and online publications on osteoporosis. Dr. Lewiecki is past-president of the International Society for Clinical Densitometry (ISCD) and current president of the Osteoporosis Foundation of New Mexico (OFNM). He is an editor for Osteoporosis International, and is on the editorial boards of the Journal of Bone and Mineral Research and the Journal of Clinical Densitometry. He has received national and international awards that include the 2021 “Dr. John P. Bilezikian ISCD Global Leadership Award” and the 2021 “Lawrence G. Raisz Memorial Lecture Award” with the Bone Health & Osteoporosis Foundation. Dr. Lewiecki is the program director of the annual Santa Fe Bone Symposium, the flagship activity of the OFNM.

Christopher Shuhart, MD, MHA, CCD
Director of the Bone Health and Osteoporosis Center
Swedish Medical Group
Dr. Shuhart is the founder and Director of the Bone Health and Osteoporosis Center at Swedish Medical Group in Seattle, Washington. He is Past-President of the International Society for Clinical Densitometry (ISCD) from 2019-2020, with multiple years in Executive and Board of Directors roles, and has served on numerous ISCD committees and task forces. He received ISCD's Paul Miller Service Award in 2022.
He is a co-author on the ISCD’s 2016 publication of “Best Practices” in the Journal of Clinical Densitometry, and was the Organizing Chair for the 2019 ISCD Position Development Conference (PDC) in Kuala Lumpur, Malaysia, as well as lead author of the 2019 PDC Executive Summary. He is the Administrative Chair for the 2023 PDC.
Dr. Shuhart received his medical degree from Dartmouth Medical School in 1987, and his Masters in Health Administration from the University of Washington in 2009. He is board certified in Family Medicine since 1990.

Statement of Independence
The ISCD maintains a policy on the use of commercial support, which ensures that all educational activities sponsored by the ISCD provide in-depth presentations that are fair, balanced, independent, and scientifically rigorous. ISCD requires faculty, planners, managers, and other individuals and their spouse/life partner who are in a position to control the content of this activity to disclose any real or apparent conflict of interest they may have as related to the content of this activity. All identified conflicts of interest are thoroughly vetted by ISCD for fair balance, scientific objectivity of studies mentioned in the materials or used as the basis for content, and appropriateness of patient care recommendations. Individual disclosures are included in the course material. No Commercial Support has been provided for this activity
Disclaimer
The information and suggestions presented at the courses, seminars and other programs sponsored by ISCD and other collaborating societies are subject to change and therefore should serve only as a foundation for further investigation and study. Any forms presented at our seminars or programs are samples only and are not necessarily authoritative. All information, procedures, and forms contained or used in such seminars or programs should serve only as a guide for use in specific situations.
Disclosure of Unlabeled Use
This educational activity may contain a discussion of published and/or investigational uses of agents that are not indicated by the FDA. ISCD does not recommend the use of any agent outside of the labeled indications. The opinions expressed in this educational activity are those of the faculty and do not necessarily represent the views of any organization associated with this activity. Please refer to the official prescribing information for each product for a discussion of approved indications, contraindications, and warnings.
Privacy & Confidentiality Statement
The ISCD will record the learner’s personal information as provided on evaluations to allow for the issuance and tracking of CME/CE certificates. The ISCD may also track aggregate responses to questions in activities and evaluations and use these data to inform the ongoing evaluation and improvement of its educational program. No individual performance data or any other personal information collected for evaluations will be shared with third parties
Attendance Cancellation Policy
All changes in registration must be made in writing to education@iscd.org. Cancellations received 30 days prior to the start of the program are eligible for a full refund minus a $50 administrative fee. Cancellations received less than 30 days prior to the start of the course will not be refunded.
Course Changes or Cancellations
ISCD reserves the right to cancel the program, change dates, how the course is taught, and/or meeting location. If a program is canceled, all program fees will be refunded unless the registered participant elects to transfer to another program. Participants will be notified of any changes within 21 days of the course start date.
-
Register
- Non-Member - $800
- Full Member - $500
- MOC Only - $800
- Community Member - $800
- Professional Member - $800
- More Information
-
This talk will review the updates on current ISCD Position guidelines 2023-2024.
Activity approved for 1.5 ASRT Category A Credits
Activity approved for up to 1.00 AMA PRA Category 1 Credits™
Activity Expires July 24, 2025Activity Description
This talk will review the updates on current ISCD Position guidelines 2023-2024.
Learning Objectives:
- Review 2023 ISCD Position Statements for Screening.
- Understand diagnosis of low bone mass/osteoporosis including VFA/Trabecular bone score and BMD reporting
- Highlight differences from 2019 positions.
Instructions:
Clinicians and Technologists are required to answer a set of online test questions following the review of the video and will have up to three attempts to answer questions correctly before losing credit for the activity. Once the activity test has been passed (75% or higher), you will be asked to evaluate the activity online and then be allowed to claim credit and print certificates.
Registration Fees
Cancellation Policy
On-Demand/ Enduring Material Cancellation
Purchases of on-demand or enduring educational materials are non-refundable.
On-Demand/Enduring Material Program Changes or Cancellations
ISCD reserves the right to cancel the program, change dates, and/or the location/method of offering. All program fees will be refunded if a program is canceled unless the registered participant elects to transfer to another program.Joint Accreditation Statement:

In support of improving patient care, this activity has been planned and implemented by Amedco LLC and ISCD. Amedco LLC is jointly accredited by the Accreditation Council for Continuing Medical Education (ACCME), the Accreditation Council for Pharmacy Education (ACPE), and the American Nurses Credentialing Center (ANCC), to provide continuing education for the healthcare team.
AMA PRA Category 1 Credit(s)™
Amedco LLC designates this live activity for a maximum of 1.00 AMA PRA Category 1 CreditsTM. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
ASRT Credit Designation
This activity has been approved by ASRT for 1.5 Category A continuing education credits.
Date of Original Release and most recent Review or Update
This activity was most recently reviewed and released on July 25, 2024.

Christopher R. Shuhart, MD, MHA, CCD
Medical Director
Swedish Bone Health and Osteoporosis
Dr. Shuhart is the founder and Director of the Bone Health and Osteoporosis Center at Swedish Medical Group in Seattle, Washington. He is Immediate Past-President of the International Society for Clinical Densitometry (ISCD) having served on the Executive Committee since 2017, on the Board of Directors since 2015 and on numerous ISCD committees and task forces. He is a co-author on the ISCD’s 2016 publication of “Best Practices” in the Journal of Clinical Densitometry, and was the Organizing Chair for the 2019 ISCD Position Development Conference in Kuala Lumpur, Malaysia.
Dr. Shuhart received his medical degree from Dartmouth Medical School in 1987, and his Masters in Health Administration from the University of Washington in 2009. He is board certified in Family Medicine since 1990.
Heenam Goel, MBBS
Endocrinologist, Reproductive Endocrinologist
CentraCare, St. Cloud, MN
Dr. Goel is an Endocrinologist at CentraCare Hospital, Saint Cloud, Minnesota and did her Endocrinology fellowship at University of Wisconsin, Madison.

Statement of Independence
The ISCD maintains a policy on the use of commercial support, which ensures that all educational activities sponsored by the ISCD provide in-depth presentations that are fair, balanced, independent, and scientifically rigorous. ISCD requires faculty, planners, managers, and other individuals and their spouse/life partner who are in a position to control the content of this activity to disclose any real or apparent conflict of interest they may have as related to the content of this activity. All identified conflicts of interest are thoroughly vetted by ISCD for fair balance, scientific objectivity of studies mentioned in the materials or used as the basis for content, and appropriateness of patient care recommendations. Individual disclosures are included in the course material. No Commercial Support has been provided for this activity
Disclaimer
The information and suggestions presented at the courses, seminars and other programs sponsored by ISCD and other collaborating societies are subject to change and therefore should serve only as a foundation for further investigation and study. Any forms presented at our seminars or programs are samples only and are not necessarily authoritative. All information, procedures, and forms contained or used in such seminars or programs should serve only as a guide for use in specific situations.
Disclosure of Unlabeled Use
This educational activity may contain a discussion of published and/or investigational uses of agents that are not indicated by the FDA. ISCD does not recommend the use of any agent outside of the labeled indications. The opinions expressed in this educational activity are those of the faculty and do not necessarily represent the views of any organization associated with this activity. Please refer to the official prescribing information for each product for discussion of approved indications, contraindications, and warnings.
Privacy & Confidentiality Statement
The ISCD will record the learner’s personal information as provided on evaluations to allow for the issuance and tracking of CME/CE certificates. The ISCD may also track aggregate responses to questions in activities and evaluations and use these data to inform the ongoing evaluation and improvement of its educational program. No individual performance data or any other personal information collected for evaluations will be shared with third parties
Attendee Cancellation Policy
All changes in registration must be made in writing to education@iscd.org. Purchase of on-demand or enduring educational materials are non-refundable.
Course Changes or Cancellations
ISCD reserves the right to cancel the program and change the dates of the offering. All program fees will be refunded if a program is canceled unless the registered participant elects to transfer to another program.
-
Register
- Non-Member - $25
- Full Member - Free!
- MOC Only - Free!
- Community Member - $25
- Professional Member - Free!
- More Information
-
The fracture risk assessment tool, FRAX ®, was released in 2008. It provides country-specific tools for estimating personalized 10-year probability of hip and major osteoporotic fracture (hip, clinical spine, distal forearm, and proximal humerus). Several adjustments have been developed that extend the range of risk factors considered by FRAX, now available through FRAXplus (www.FRAXplus.org). Meanwhile, there is ongoing research that will inform the next iteration of the FRAX tool.
Activity approved for 1.25 ASRT Category A Credits
Activity approved for up to 1.0 AMA PRA Category 1 Credits™
Activity Expires July 24, 2025Activity Description
The fracture risk assessment tool, FRAX ®, was released in 2008. It provides country-specific tools for estimating personalized 10-year probability of hip and major osteoporotic fracture (hip, clinical spine, distal forearm, and proximal humerus). Several adjustments have been developed that extend the range of risk factors considered by FRAX, now available through FRAXplus (www.FRAXplus.org). Meanwhile, there is ongoing research that will inform the next iteration of the FRAX tool.
Learning Objectives:
- Summarize the development of the FRAX tool, including its strength and limitations.
- Understand adjustments to FRAX that permit incorporation of additional risk factor information.
- Explain the clinical impact and application of FRAXplus and, eventually, FRAX-2 on clinical decision-making.
Instructions:
Clinicians and Technologists are required to answer a set of online test questions following the review of the video and will have up to three attempts to answer questions correctly before losing credit for the activity. Once the activity test has been passed (75% or higher), you will be asked to evaluate the activity online and then be allowed to claim credit and print certificates.
Registration Fees
Cancellation Policy
On-Demand/ Enduring Material Cancellation
Purchases of on-demand or enduring educational materials are non-refundable.
On-Demand/Enduring Material Program Changes or Cancellations
ISCD reserves the right to cancel the program, change dates, and/or the location/method of offering. All program fees will be refunded if a program is canceled unless the registered participant elects to transfer to another program.Joint Accreditation Statement:

In support of improving patient care, this activity has been planned and implemented by Amedco LLC and ISCD. Amedco LLC is jointly accredited by the Accreditation Council for Continuing Medical Education (ACCME), the Accreditation Council for Pharmacy Education (ACPE), and the American Nurses Credentialing Center (ANCC), to provide continuing education for the healthcare team.
AMA PRA Category 1 Credit(s)™
Amedco LLC designates this live activity for a maximum of 1.00 AMA PRA Category 1 CreditsTM. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
ASRT Credit Designation
This activity has been approved by ASRT for 1.25 Category A continuing education credits.
Date of Original Release and most recent Review or Update
This activity was most recently reviewed and released on July 25, 2024.

William Leslie, MD MSc FRCPC
Dr. Leslie is Professor of Medicine and Radiology at the University of Manitoba with over 550 peer-reviewed publications. His research interests are in fracture risk assessment, osteoporosis testing and nuclear diagnostic techniques. He is Past President of the International Society for Clinical Densitometry, Past Chair of the Osteoporosis Canada Scientific Advisory Council, and Director of the Manitoba Bone Density Program.

Statement of Independence
The ISCD maintains a policy on the use of commercial support, which ensures that all educational activities sponsored by the ISCD provide in-depth presentations that are fair, balanced, independent, and scientifically rigorous. ISCD requires faculty, planners, managers, and other individuals and their spouse/life partner who are in a position to control the content of this activity to disclose any real or apparent conflict of interest they may have as related to the content of this activity. All identified conflicts of interest are thoroughly vetted by ISCD for fair balance, scientific objectivity of studies mentioned in the materials or used as the basis for content, and appropriateness of patient care recommendations. Individual disclosures are included in the course material. No Commercial Support has been provided for this activity
Disclaimer
The information and suggestions presented at the courses, seminars and other programs sponsored by ISCD and other collaborating societies are subject to change and therefore should serve only as a foundation for further investigation and study. Any forms presented at our seminars or programs are samples only and are not necessarily authoritative. All information, procedures, and forms contained or used in such seminars or programs should serve only as a guide for use in specific situations.
Disclosure of Unlabeled Use
This educational activity may contain a discussion of published and/or investigational uses of agents that are not indicated by the FDA. ISCD does not recommend the use of any agent outside of the labeled indications. The opinions expressed in this educational activity are those of the faculty and do not necessarily represent the views of any organization associated with this activity. Please refer to the official prescribing information for each product for discussion of approved indications, contraindications, and warnings.
Privacy & Confidentiality Statement
The ISCD will record the learner’s personal information as provided on evaluations to allow for the issuance and tracking of CME/CE certificates. The ISCD may also track aggregate responses to questions in activities and evaluations and use these data to inform the ongoing evaluation and improvement of its educational program. No individual performance data or any other personal information collected for evaluations will be shared with third parties
Attendee Cancellation Policy
All changes in registration must be made in writing to education@iscd.org. Purchase of on-demand or enduring educational materials are non-refundable.
Course Changes or Cancellations
ISCD reserves the right to cancel the program and change the dates of the offering. All program fees will be refunded if a program is canceled unless the registered participant elects to transfer to another program.
-
Register
- Non-Member - $25
- Full Member - Free!
- MOC Only - Free!
- Community Member - $25
- Professional Member - Free!
- More Information
-
This course will focus on the future outlook for measuring bone health and fracture risk prediction. We will utilize data on the clinical utility and available evidence supporting these modalities.
Activity approved for 1.5 ASRT Category A Credits
Activity approved for up to 1.0 AMA PRA Category 1 Credits™
Activity Expires July 24, 2025Activity Description
This course will focus on the future outlook for measuring bone health and fracture risk prediction. We will utilize data on the clinical utility and available evidence supporting these modalities.
Learning Objectives:
- Summarize current tools for assessing bone health and fracture risk.
- Identify evolving options for assessing bone health and fracture risk.
- Examine data on the clinical utility and available evidence supporting these modalities.
Instructions:
Clinicians and Technologists are required to answer a set of online test questions following the review of the video and will have up to three attempts to answer questions correctly before losing credit for the activity. Once the activity test has been passed (75% or higher), you will be asked to evaluate the activity online and then be allowed to claim credit and print certificates.
Registration Fees
Cancellation Policy
On-Demand/ Enduring Material Cancellation
Purchases of on-demand or enduring educational materials are non-refundable.
On-Demand/Enduring Material Program Changes or Cancellations
ISCD reserves the right to cancel the program, change dates, and/or the location/method of offering. All program fees will be refunded if a program is canceled unless the registered participant elects to transfer to another program.Joint Accreditation Statement:

In support of improving patient care, this activity has been planned and implemented by Amedco LLC and ISCD. Amedco LLC is jointly accredited by the Accreditation Council for Continuing Medical Education (ACCME), the Accreditation Council for Pharmacy Education (ACPE), and the American Nurses Credentialing Center (ANCC), to provide continuing education for the healthcare team.
AMA PRA Category 1 Credit(s)™
Amedco LLC designates this live activity for a maximum of 1.00 AMA PRA Category 1 CreditsTM. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
ASRT Credit Designation
This activity has been approved by ASRT for 1.5 Category A continuing education credits.
Date of Original Release and most recent Review or Update
This activity was most recently reviewed and released on July 25, 2024.

S. Bobo Tanner, MD, CCD
Director of the Vanderbilt Osteoporosis Clinic
Vanderbilt University Medical Center
S. Bobo Tanner, MD, CCD, is a graduate of Harvard University in Cambridge, Massachusetts, with a BA in History and Science, and received his medical degree with honors from Bowman Gray Medical School of Wake Forest University, Winston-Salem, North Carolina. He pursued his internship and residency at the Vanderbilt University Hospital in Nashville, Tennessee, and completed fellowships and board certification in Rheumatology and in Allergy/Immunology. Dr. Tanner served as a Registrar in the Department of Rheumatology at the Royal Free Hospital, London, England.
Afterwards he returned to Vanderbilt and was Founder and Director of the Vanderbilt Osteoporosis Clinic which is part of the Rheumatology Division.
In 2007 he received the Clinician of the Year Award from International Society for Clinical Densitometry and in 2017 he was recognized with the President’s Award from the Tennessee Rheumatology Society.
His professional membership and service includes the American College of Physicians, Fellow of the American College of Rheumatology, member of the Royal Society of Medicine, and the American Academy of Asthma, Allergy & Immunology, past secretary/ treasurer of the Tennessee Rheumatology Society, past president of the Tennessee Allergy Society and past president of the International Society for Clinical Densitometry. He currently works on the ISCD Education Council where he updates and teaches the CME Bone Density Courses throughout the year. He is a Certified Clinical Densitometrist. His is on the editorial board for the Journal of Clinical Densitometry and Therapeutic Advances in Musculoskeletal Disease.

Statement of Independence
The ISCD maintains a policy on the use of commercial support, which ensures that all educational activities sponsored by the ISCD provide in-depth presentations that are fair, balanced, independent, and scientifically rigorous. ISCD requires faculty, planners, managers, and other individuals and their spouse/life partner who are in a position to control the content of this activity to disclose any real or apparent conflict of interest they may have as related to the content of this activity. All identified conflicts of interest are thoroughly vetted by ISCD for fair balance, scientific objectivity of studies mentioned in the materials or used as the basis for content, and appropriateness of patient care recommendations. Individual disclosures are included in the course material. No Commercial Support has been provided for this activity
Disclaimer
The information and suggestions presented at the courses, seminars and other programs sponsored by ISCD and other collaborating societies are subject to change and therefore should serve only as a foundation for further investigation and study. Any forms presented at our seminars or programs are samples only and are not necessarily authoritative. All information, procedures, and forms contained or used in such seminars or programs should serve only as a guide for use in specific situations.
Disclosure of Unlabeled Use
This educational activity may contain a discussion of published and/or investigational uses of agents that are not indicated by the FDA. ISCD does not recommend the use of any agent outside of the labeled indications. The opinions expressed in this educational activity are those of the faculty and do not necessarily represent the views of any organization associated with this activity. Please refer to the official prescribing information for each product for discussion of approved indications, contraindications, and warnings.
Privacy & Confidentiality Statement
The ISCD will record the learner’s personal information as provided on evaluations to allow for the issuance and tracking of CME/CE certificates. The ISCD may also track aggregate responses to questions in activities and evaluations and use these data to inform the ongoing evaluation and improvement of its educational program. No individual performance data or any other personal information collected for evaluations will be shared with third parties
Attendee Cancellation Policy
All changes in registration must be made in writing to education@iscd.org. Purchase of on-demand or enduring educational materials are non-refundable.
Course Changes or Cancellations
ISCD reserves the right to cancel the program and change the dates of the offering. All program fees will be refunded if a program is canceled unless the registered participant elects to transfer to another program.
-
Register
- Non-Member - $25
- Full Member - Free!
- MOC Only - Free!
- Community Member - $25
- Professional Member - Free!
- More Information
